The FDA approved Cobenfy
Schizophrenia is a complex and debilitating mental disorder characterized by a range of symptoms, including hallucinations, delusions, cognitive deficits, and emotional dysregulation. Despite advancements in antipsychotic medications, many patients experience incomplete symptom relief and significant side effects. As a result, there is a growing interest in alternative therapeutic targets, including the muscarinic acetylcholine receptors (mAChRs).
Muscarinic Acetylcholine Receptors (mAChRs)
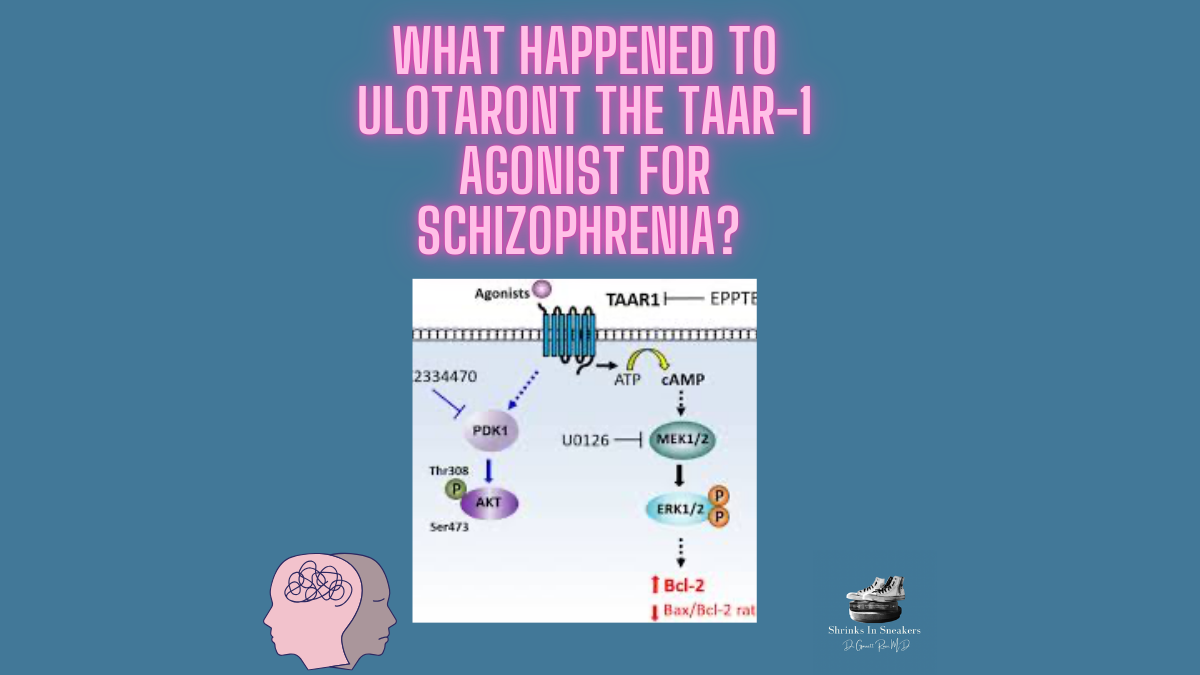
The mAChRs are G protein-coupled receptors involved in various central nervous system functions, including cognition, learning, memory, and mood regulation. There are five subtypes of mAChRs (M1-M5), with the M1, M2, M3, and M4 subtypes playing significant roles in modulating neural activity related to schizophrenia.
M1 Muscarinic Agonists
The M1 receptor is primarily expressed in the cortex and hippocampus, regions crucial for cognitive processing. M1 agonists have shown promise in improving cognitive deficits and reducing psychotic symptoms in schizophrenia. Research indicates that M1 activation can enhance cholinergic neurotransmission and modulate glutamate and dopamine systems, potentially alleviating both positive and negative symptoms.
M2 Muscarinic Agonists
M2 receptors are predominantly found in the basal forebrain and play a role in modulating acetylcholine release. Although less studied than M1, M2 agonists may help balance neurotransmitter release, contributing to improved cognitive function and reduced psychotic symptoms.
M3 Muscarinic Agonists
The role of M3 receptors in schizophrenia is not as well understood as M1 and M4 receptors. However, M3 receptors are involved in various physiological processes, including insulin secretion and smooth muscle contraction. Research is ongoing to determine their potential therapeutic benefits in schizophrenia.
M4 Muscarinic Agonists
M4 receptors are highly expressed in the striatum, a brain region implicated in the regulation of motor control and reward processing. M4 agonists have shown potential in reducing dopaminergic hyperactivity, which is associated with positive symptoms of schizophrenia, such as hallucinations and delusions. Additionally, M4 activation may help mitigate side effects associated with conventional antipsychotics, such as extrapyramidal symptoms.
Clinical Implications and Future Directions
The therapeutic potential of M1-M4 muscarinic agonists in schizophrenia is an exciting area of research. Targeting these receptors may offer a novel approach to address the cognitive and negative symptoms of schizophrenia, which are often resistant to current treatments. Ongoing clinical trials and preclinical studies are crucial to understanding the efficacy, safety, and mechanisms of action of these compounds.
Conclusion The exploration of M1-M4 muscarinic agonists represents a promising frontier in the treatment of schizophrenia. By modulating cholinergic, glutamatergic, and dopaminergic systems, these agents have the potential to provide more comprehensive symptom relief with fewer side effects compared to traditional antipsychotics. Continued research and development are essential to bring these innovative treatments to clinical practice, offering hope for improved outcomes for individuals with schizophrenia.